Bone marrow edema (BME) is cropping up in MRI image reports and can be harbingers of subsequent cartilage loss where it occurs – though some can progress to frank bone osteonecrosis. Clinically, the presence of knee effusion make BME 77.9% likely.
Association of Knee Effusion Detected by Physical Examination With Bone Marrow Lesions: Cross-Sectional and Longitudinal Analyses of a Population- Based Cohort
Jolanda Cibere et al
Arthritis Care & Research
Vol. 71, No. 1, January 2019, pp 39–45
https://onlinelibrary.wiley.com/doi/abs/10.1002/acr.23575
There is talk of maybe been enlisting victims in a Bone marrow edema study but there are none active right now:
https://clinicaltrials.gov/ct2/results?cond=Bone+Marrow+Edema+Syndrome&term=&cntry=&state=&city=&dist=
Possible Treatments:
Strontium Ranelate -so so
Pelletier, Jean-Pierre, et al.
Disease-modifying effect of strontium ranelate in a subset of patients from the Phase III knee osteoarthritis study SEKOIA using quantitative MRI: reduction in bone marrow lesions protects against cartilage loss.
Annals of the rheumatic diseases 74.2 (2015): 422-429.
http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.828.7608&rep=rep1&type=pdf
- No Canadian DIN number yet study was done in Quebec, Canada so suspect must have been compounded.
- “SrRan” was found to reduce bone resorption and increase formation
strontium ranelate was given at 2 gm/day for 2-3 years from the looks of it. - “significantly reducing CVL (cartilage volume loss) in the plateau and BML (bone marrow lesion) progression in the medial compartment”
- Pain levels were different from onset :SrRan 44.5±20.0/100 vs Placebo 38.5±21.4. so made analysis more difficult.
- Results on pain were so poor they did not publish them except to say they will able to show something on multivariate regression though said “data not shown” – tells me it has no clinical relevance pain-wise – they did admit the did not achieve some of their primary objectives
Iloprost – not really
Mayerhoefer, M. E., et al.
Short-term outcome of painful bone marrow oedema of the knee following oral treatment with iloprost or tramadol: results of an exploratory phase II study of 41 patients.
Rheumatology 46.9 (2007): 1460-1465.
https://academic.oup.com/rheumatology/article/46/9/1460/1790006
- No Canadian DIN number
- 50 µg of iloprost clathrate (Schering AG, Berlin, Germany) three times daily on days 1–3; after day 3, individual dose adjustment was allowed (range: 50 µg twice daily up to 2 × 50 µg three times daily).
- The oral extended-release formulation of iloprost was developed to mimic the plasma concentration time curve of intravenous iloprost during a 6 h infusion at a dose rate of 1.5 ng/kg/min.
- Conclusion- The analgesic effect of iloprost and tramadol was similar. BME regression on MR images was more pronounced under iloprost treatment.
Zoledronic Acid – no
Laslett, Laura Louise, et al.
Zoledronic acid reduces knee pain and bone marrow lesions over 1 year: a randomised controlled trial.
Annals of the rheumatic diseases 71.8 (2012): 1322-1328.
https://ard.bmj.com/content/71/8/1322.short
- aged 50–80 years (n=59)
- clinical knee osteoarthritis and knee BML (bone marrow edema)
- Single infusion of Zoledronic acid 5 mg/100 mlResults:
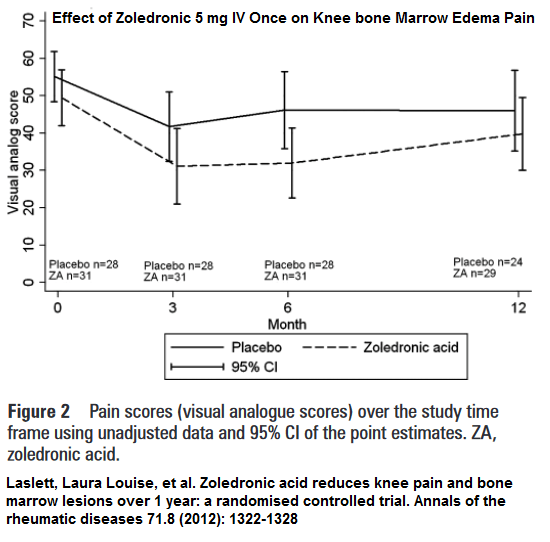
- No Canadian DIN number
- Results were mediocre and standard deviation bars are not apart so I see no real significant difference
- I call this a MORON study – as seen from pamidronate study below, and previous pamidronate studies on compression fractures of spine, it took THREE infusions to get good results – I also call them ONE-SHOT-WONDERS
so possible could have got results with THREE infusions
a repeat of this study was done and showed No benefit
Zoledronic acid infusions had no effect on knee osteoarthritis with bone marrow edema lesion:
Cai, G., et al.
OP0016 A multicentre randomised controlled trial of zoledronic acid for osteoarthritis of the knee with bone marrow lesions.
(2018): 57-58.
https://ard.bmj.com/content/77/Suppl_2/57.2
- 5 mg/100 mls iv yearly
no benefit
In actual knee osteonecrosis:
Kraenzlin, Marius E., et al.
Possible beneficial effect of bisphosphonates in osteonecrosis of the knee.
Knee surgery, sports traumatology, arthroscopy 18.12 (2010): 1638-1644.
http://doc.rero.ch/record/319972/files/167_2010_Article_1106.pdf
- pamidronate 120 mg i.v. divided in 3–4 perfusions over 2 weeks
- followed by oral bisphosphonate treatment with alendronate 70 mg weekly for 4–6 months.
- results in pain relief : “VAS decreasing from 8.2 ± 1.2 at baseline to 5.02 ± 0.6 after 4–6 weeks (P<.001). After 6 months, the VAS decreased by 80% ( p <0.001).”
Not all studies were positive:
Meier, C., et al.
Effect of ibandronate on spontaneous osteonecrosis of the knee: a randomized, double-blind, placebo-controlled trial.
Osteoporosis international 25.1 (2014): 359-366.
https://link.springer.com/article/10.1007/s00198-013-2581-5
- “participants were randomized to receive 13.5 mg ibandronate (Bonviva IV; Roche Pharma AG, Switzerland) or placebo intravenously divided in four injections within
2 weeks (once 1.5 mg then 3 mg per injection) and followed by a fifth injection after 3 months (3 mg).” - “no beneficial effect over and above anti-inflammatory medication”
For patellar bone marrow edema, the The Formfit Knee Hinged Lateral J support showed benefit after 6 weeks:
Callaghan, Michael J., et al.
A randomised trial of a brace for patellofemoral osteoarthritis targeting knee pain and bone marrow lesions.”
Annals of the rheumatic diseases 74.6 (2015): 1164-1170.
https://ard.bmj.com/content/74/6/1164
Results:
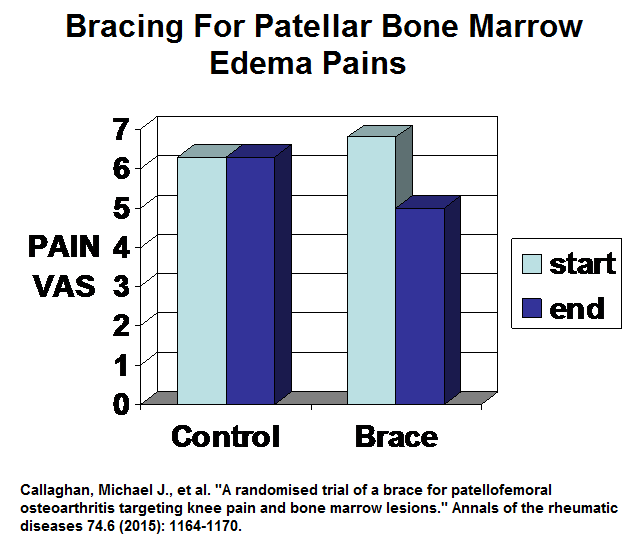
Well, not going to be running any time soon with bracing…
Biphosphonates on OA
Interestingly biphosphonates were found to help knee OA pains in first 3 years of use:
Laslett, Laura L., et al.
Effect of bisphosphonate use in patients with symptomatic and radiographic knee osteoarthritis: data from the Osteoarthritis Initiative.
Annals of the rheumatic diseases 73.5 (2014): 824-830.
http://eprints.whiterose.ac.uk/89937/3/Laslett%20et%20al_manuscript%20including%20figures.pdf
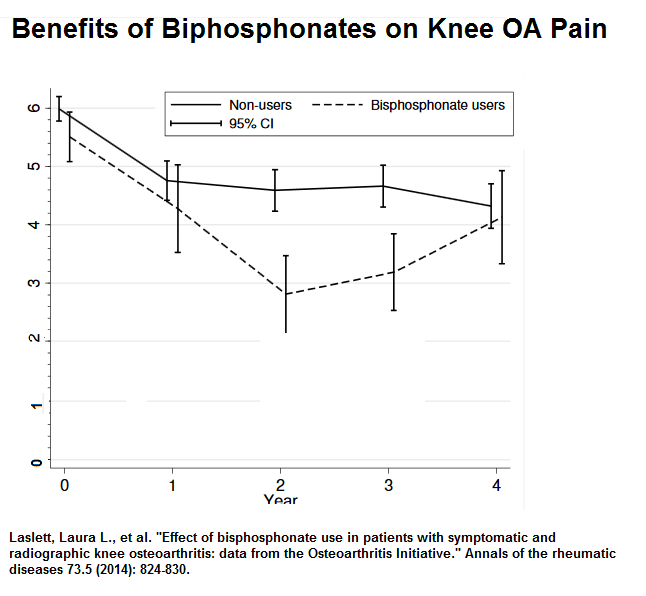
Alendronate 35 mg/wk reduced hip OA pains but failed to help AO pregression:
Nishii, Takashi, et al.
Alendronate treatment for hip osteoarthritis: prospective randomized 2-year trial.
Clinical rheumatology 32.12 (2013): 1759-1766.
https://link.springer.com/article/10.1007/s10067-013-2338-8
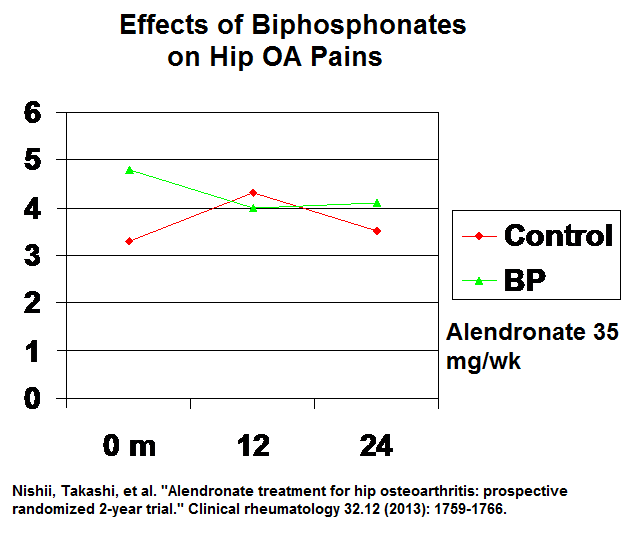
I can’t make any sense of that study – subjects were not matched for pain at start and it went downhill from there.
Comment – up until now I have not written up bone marrow edema as no treatment seemed worth writing up. I am forced to admit it might not have changed much